
ALL MATERIAL COPYRIGHT KEVIN SCOTT 2011. LINKS TO THIS SITE ARE WELCOME BUT DO NOT COPY MATERIAL FROM THIS SITE TO ANY OTHER WEBPAGE.
If you find this site useful, please support it by making a donation of $1 to help maintain and develop it. Click on the PAYPAL DONATE button to do this safely. But there is no obligation - please avail yourself of the information and facilities of the site at no charge.

Hypericum Perforatum, St John's Wort is very easily cultivated. Grown from seed in compost in a standard seed tray, the young plants are robust and grow well in temperate conditions. Replanted in an earthenware tub they rapidly advance in size and adventitiously in extent until the tub is filled with the plant.It has been reported that St Joh's Wort does not do well when grown in tubs, but experience reported here does not confirm that. Conversely, non-confined cultivation is not generally recommended, as St John's Wort tends to become a pest, spreading rapidy and potentially being a hazard to grazing animals.





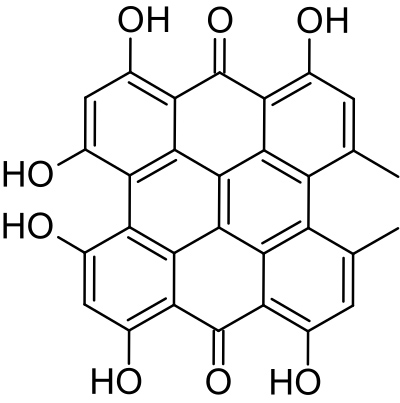
The molecular structure of hypericin is shown to the right. In its free form it is a deep cherry red colour in organic solvents and forms blue-black needles when crystallised from pyrindine +methanolic HCl. The Merck Index records hypericin to be almost insoluble in most organic solvents, but, while insoluble in acetonitrile, this study found it to be very soluble in ethanol. It has also been reported as soluble in acetone/ethanol mixtures which can be used to extract hypericin from the plant.
Hypericin shows two peaks in the UV-vis spectrum, one at 590 nm (Extinction Coefficient (M-1 cm-1): 4.4 104 and another at about 480nm.
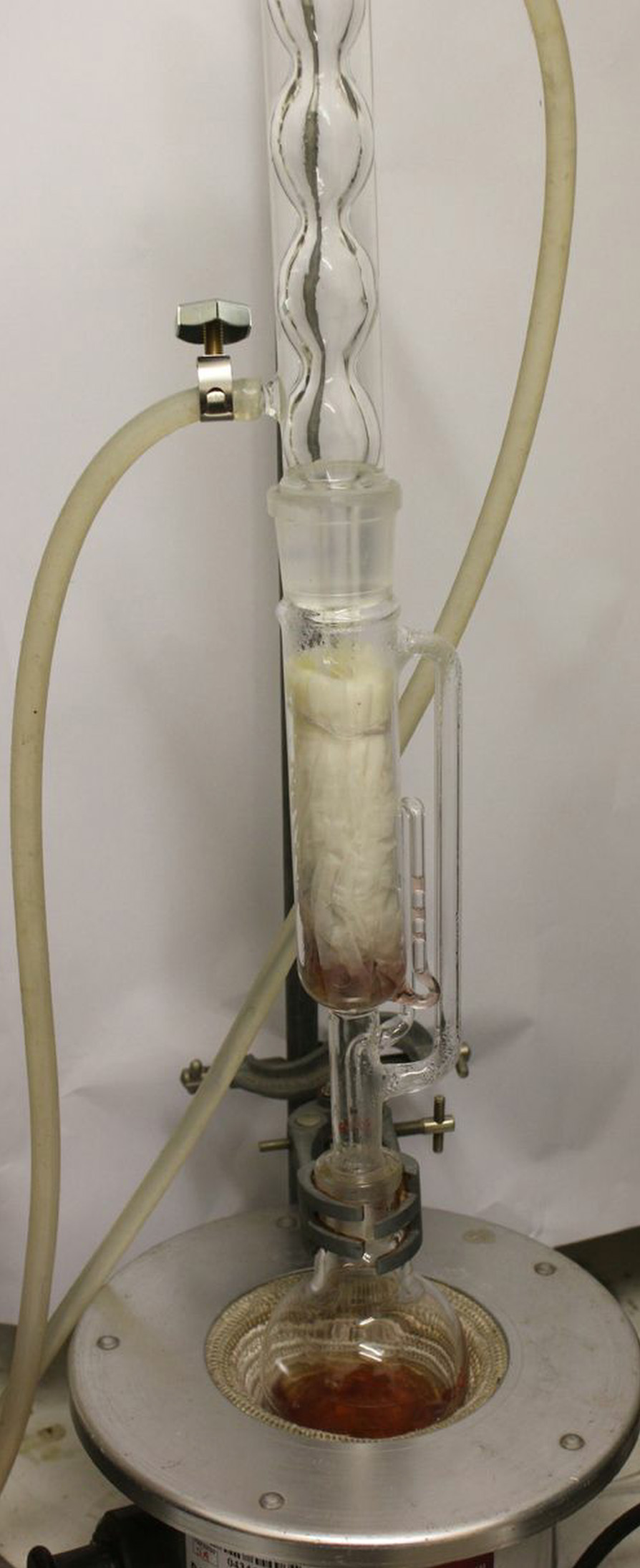

Several methods of extraction have been tried in this study:
Extraction by trituration with vodka in a mortar followed by filtration: This resulted in a red solution of hypericin in alcohol/water mixture, but this rapidly decolorised and formed a brown precipitate.
Soxhlet extraction using rectified spirit: This produced a deep red extract, which was stable.
Other solvents: preliminary efforts to extract hypericin by trituration of the flowers with diethyl ether and in acetonitrile were unsuccessful.
Trituration with distilled water in a mortar: This resulted in a red solution of hypericin in water whch turned yellow upon the addition of sodium carbonate and red once again when the latter was partially neutralised with sulphuric acid.
A Sodium carbonate/sodium bicarbonate buffer was prepared by dissolving 0.577 gm NaHCO3 and 0.07gm Na2CO3 in 250ml distilled water. 20 ml of this bufer solution was used to make an extraction of 2.83 gms of St John's Wort flowers by trituration in a mortar. a red solution was obtained which turned brown on standing. It appeared to be a colloidal suspension.
Ultimately a soxhlet extraction using rectified spirit (95% Ethanol, 5% Water) was used to extract the hypericin. 10 -12 gms of freshly harvested flowers, buds and spent blooms were packed into a soxhlet thimble and the latter installed in a 60ml soxhlet extractor and assembled with a condenser and 125 ml flask into which 100 ml 95% Ethanol had been introduced. The flask was heated with an electric mantle and after about 20 soxhlet extraction cycles, the liquid in the flask became deep red and little further extraction was apparently taking place. The extract and cooled and refridgerated.
Clearly the best way to assess the concentration of hypericin in the extracts would be to use HPLC with a standard hypericin sample, or use UV-vis spectrophotometry with the extinction data above. However, in the absence of these techniques a simple colorimetric estimate was employed.
A single commercial pill of St John's Wort extract which was asserted by the manufacturer to contain 1 mg of Hypericin was ground in a mortar and extracted with 10 ml of 1.6M Sulphuric acid. After centrifuging, this resulted in a pink supernatant liquid, the colour intensity of which was compared visually with successively diluted samples of the Hypericin solution obtained by soxhlet extraction. This allowed an approximate estimate of the Hypericin concentration in the extracted solutions. This process gave an estimated concentration of 2.5 mg /ml +/- 1 mg/ml.
The dosage suggested on the packaging of commercial St John's Wort pills is 1 QD, but some physicians who suggest the use of hypericin for treating mild to moderate depression, suggest 6 mg QD.
In the case under study in this report, a does of 10 mg QD for three days appeared to dispel moderate depression very rapidly, and when the condition which began to return on cessation of the treatment it was once again dispelled by a further similar dose. There was, of course, no control group, no blind testing and no accounting for the placebo effectin this series of events. It can only be said that it might have worked!